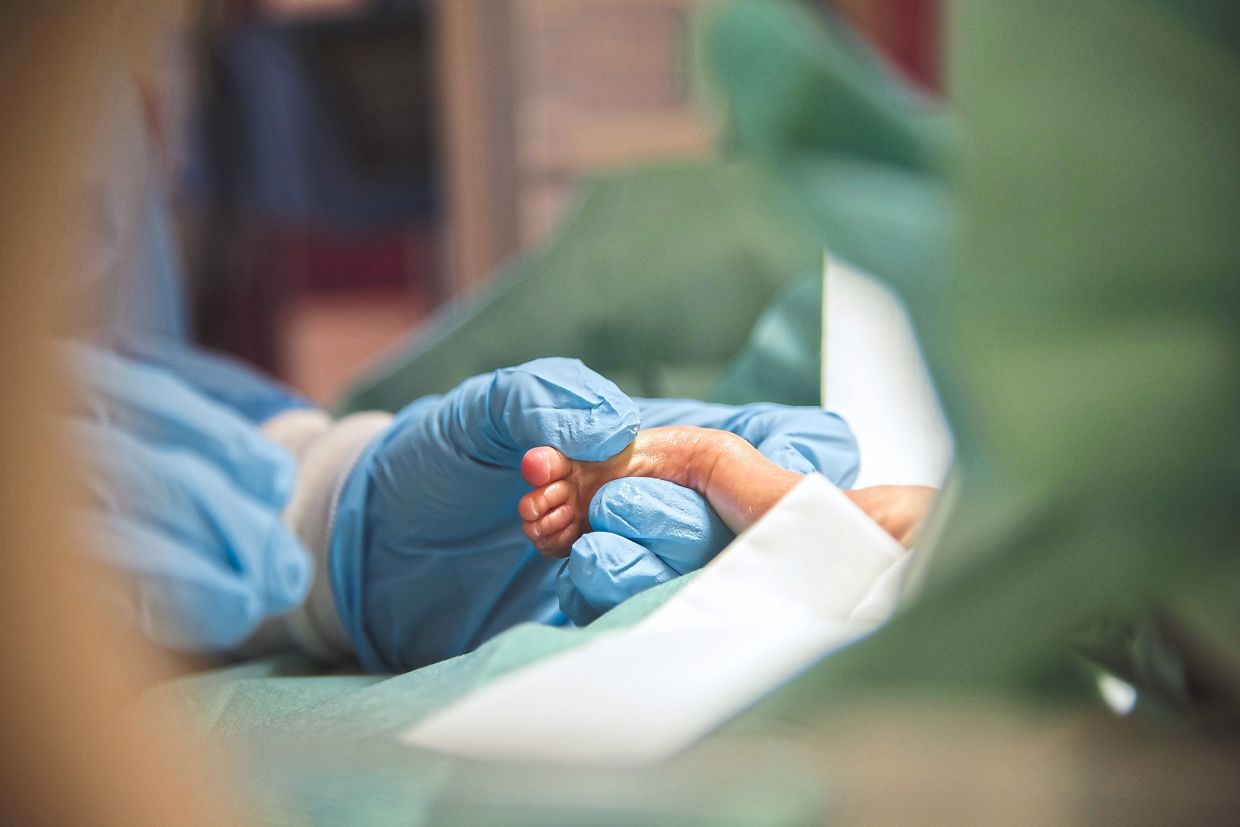
The objective of newborn screening (NBS) is the identification of serious and/or rare conditions that can affect a newborn’s future health and survival.
Many of these conditions – e.g.
inborn errors of metabolism, endocrine diseases, haemoglobinopathies, spinal muscular atrophy, congenital heart disease and hearing loss – may not have clinically-detectable features at birth, thus hampering early detection that is vital for effective treatment and better outcomes.
NBS is a public health programme that helps to reduce the medical costs associated with serious and rare diseases by screening the entire population.
The rationale for NBS is based on the economic principle that screening all newborns and managing cases before they become symptomatic is more cost-effective than treating symptomatic individuals.
The economic benefits of NBS have been established in many countries.
What it involves NBS is performed in the first 24 to 48 hours of life, and involves: Taking a few drops of blood from the baby’s heel and sending it to the laboratory, i.e.
a heel prick test or newborn blood spot screeningA hearing screen to ascertain how the baby responds to sound, andPulse oximetry that measures blood oxygen levels to detect critical congenital heart disease.
The blood spot screening of many countries, particularly developed ones, covers: Metabolic conditions, e.g.
phenylketonuria, maple syrup urine diseaseHormonal conditions, e.g.
congenital hypothyroidism, congenital adrenal hyperplasiaHaematological (blood) conditions, e.g.
haemoglobinopathy, thalassaemia, sickle cell diseaseRare conditions, e.g.
spinal muscular atrophy (SMA).
Screening the genes The human body is made up of cells, each of which contains genetic information in a code called DNA (deoxyribonucleic acid), which makes everyone unique.
DNA works like a computer programme that instructs each cell in the body on what to do and how to do it.
The genome is a complete set of these instructions.
Science has now developed sufficiently to make it possible for this DNA information to be read to help the understanding of disease.
Sometimes, DNA changes can be inherited and passed down to family members.
These changes can mean that a person has a greater risk of developing certain conditions.
The use of newborn genomic screening (NGS) is a significant advancement in neonatal health assessment, foreshadowing a new era of early disease detection and prevention.
Until recently, population-based detection of treatable newborn disorders has been limited to metabolic and endocrine diseases, except for SMA.
This limitation has been due to technical constraints, because traditional screening methods rely on detecting excess metabolites.
On the other hand, NGS offers the possibility of substantially expanding NBS to include a wider range of treatable genetic conditions that would otherwise be undetected.
NGS has varied in different countries, with some implementing it at the population level and others in specific settings.
The initial findings of NGS reflect its feasibility and the potential to revolutionise neonatal care.
However, there are also challenges in implementation.
The diagnosis of many genetic conditions have been facilitated by technological advances in recent years.
In addition, some genetic conditions that have previously been untreatable have, in recent years, become treatable.
An example is SMA.
Spinal muscular atrophy SMA is a genetic condition involving the failure of nerves in the spinal cord to transmit messages from the brain to muscles.
The muscles then become damaged and weak, and eventually, waste away (i.e.
SMA affects all body muscles.
Muscles in the shoulders, hips and back are often the most severely affected, as well as muscles for feeding and swallowing, and those involved in breathing and coughing, resulting in increased likelihood of pneumonia and other lung problems.....